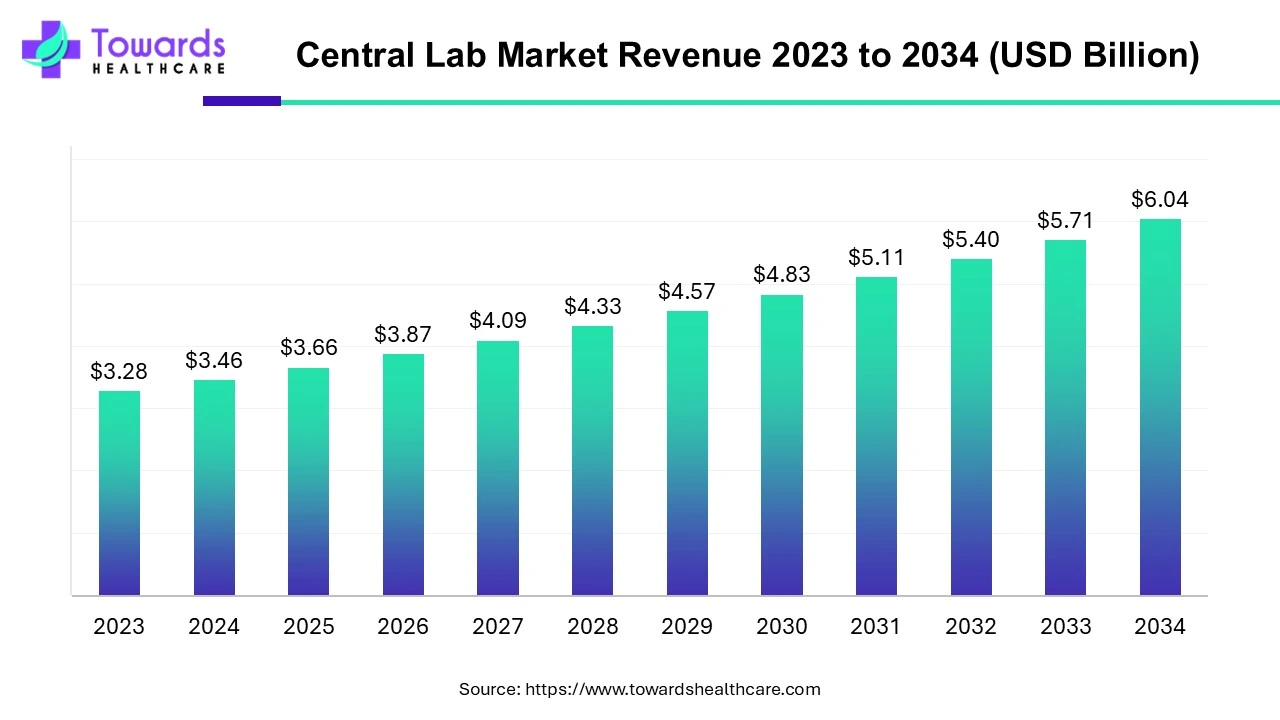
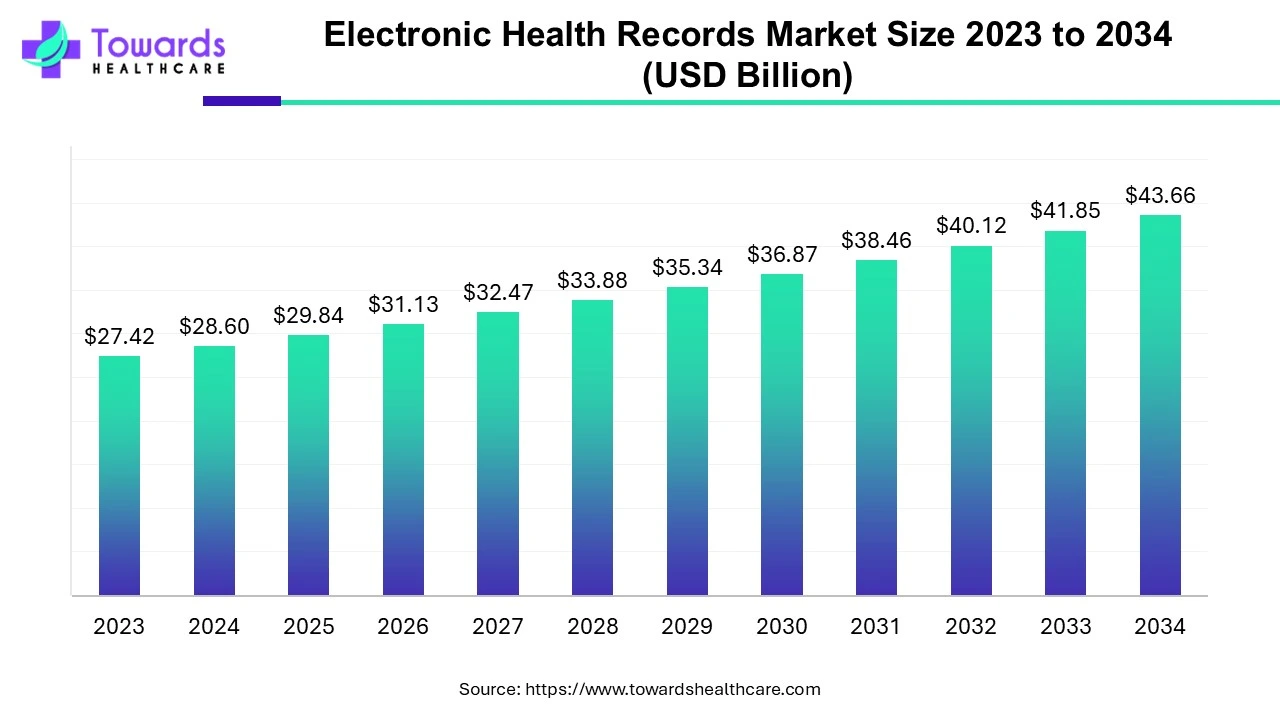
The global Electronic Health Records (EHR) market was USD 28.60 billion in 2024 and is projected to reach USD 43.66 billion by 2034, growing at a CAGR of 4.32% as healthcare systems digitalize, cloud solutions scale, and data-driven care expands.
Download the free sample and get the complete insights and forecasts report on this market @ https://www.towardshealthcare.com/download-sample/5103
Market Size in Electronic Health Records (EHR) Market
1. Absolute market value (2024)
USD 28.60 billion: represents total estimated revenue across software (EHR platforms), services (implementation, training, support), and associated infrastructure (hosting, integrations) in 2024.
Implication: This is the commercial scale reflecting vendor sales, subscription revenue, professional services, and ancillary revenues (e.g., integrations, analytics modules).
2. Forecast value (2034)
USD 43.66 billion by 2034: implies absolute growth of USD 15.06 billion over the decade.
Implication: Continued, but modest, market expansion—largely from upgrades, cloud migration, geographic expansion, and added-value services (analytics, AI modules, interoperability services).
3. CAGR (2024–2034)
4.32% CAGR: a steady moderate growth rate consistent with enterprise healthcare IT adoption curves—slower than early-stage booms but sustained by regulation and digital transformation.
Implication: Mature market with incremental adoption (replacements and upgrades) rather than explosive new purchases.
4. Market composition by offering (derived)
Software (core EHR): licensing/subscriptions for EHR modules — clinical, administrative, revenue-cycle.
Services: implementation, integration (HIS, lab, pharmacy), training, managed services.
Cloud hosting & infrastructure: growing portion as web server/cloud-based models increase.
5. 2023 structural datapoints affecting size
Cloud-based (48% share in 2023): nearly half the market by revenue/use-case, indicating major shift from on-prem to cloud-hosted EHRs.
Clinical applications (46% share in 2023): clinical modules (notes, order entry, medication management) are nearly half the market — indicative that clinical function remains core revenue driver.
6. Segment-level revenue trajectory (by type: acute/post-acute/ambulatory)
Revenue numbers by year (2024–2034) show acute as the largest single type in absolute terms, with post-acute demonstrating above-average growth (percentage terms) — this implies both continued hospital investment and increasing decentralization to post-acute/home care.
7. Unit economics and pricing pressure
As cloud subscription models dominate, pricing moves from large upfront capital expenditure (CapEx) to recurring operational expenditure (OpEx) — affects customer procurement cycles and vendor revenue recognition.
8. Market fragmentation and concentration
Large vendors hold substantial hospital footprints (enterprise sales), while medium/small vendors and open-source systems capture ambulatory and cost-sensitive markets — contributing to a two-tier market structure.
Market Trends in Electronic Health Records (EHR) Market
1. Increasing number of hospitalizations → adoption impetus
Mechanism: Higher inpatient volumes increase demand for real-time record management, clinician coordination, bed and throughput management modules.
Impact: Hospitals prioritize EHR features for inpatient workflows (CPOE, ICU charting, medication administration).
2. Demand for improved patient care → workflow optimization
How EHRs help: Standardized order sets, integrated decision support, allergy/medication reconciliation, longitudinal patient views.
Outcome measures: Reduced duplicate tests, fewer medication errors, improved adherence to clinical guidelines.
3. Technological advancements fueling new applications
AI/analytics integration: Predictive risk scores, sepsis alerts, readmission risk.
mHealth & telehealth: EHRs connecting with telemedicine sessions and remote monitoring.
Interoperability protocols (implicit): APIs, standard message formats embed into EHR roadmaps.
4. Rising investments in IT & telecom infrastructure
Public & private spending: Governments incentivize EHR adoption; private health systems invest to meet digital transformation targets.
Effect: Better broadband and data centers enable cloud adoption and distributed care models.
5. Cloud-based dominance (48% share in 2023)
Drivers: lower maintenance burden, faster upgrades, scalability for analytics.
Operational impact: Vendors shift focus to SaaS models; customers trade CapEx for OpEx.
6. Clinical applications dominance (46% share in 2023)
Focus areas: documentation, clinical decision support, CPOE, order/result tracking.
Reasoning: Clinical modules directly affect patient safety and outcomes—hence prioritized budgets.
7. Policy & regulation (examples provided)
HHS initiatives (Jan 2024): push on patient access and interoperability guidelines.
Germany’s Patient Data Protection Act: shows legal compulsion to adopt electronic records in some jurisdictions — accelerates regional uptake.
8. Historical computer-based records adoption trend
2009 → 2019: 46% to 88% adoption indicates rapid digital shift; inertia for the remaining segments likely due to resource limits or complexity.
9. Patient engagement trend
Patient portals & MyChart-style access: drives demand for patient-facing features, secure messaging, access controls, and consent management.
10. Data security & privacy
Priority: encryption, role-based access, audit trails, regulatory compliance (e.g., HIPAA-like regimes).
Market effect: vendors invest in security modules — increases product differentiation and pricing for secure offerings.
Role of Artificial Intelligence
1. Predictive risk stratification (readmission, deterioration)
What it does: Models analyze longitudinal EHR data (labs, vitals, comorbidities) to forecast risk of readmission, ICU transfer or deterioration.
EHR integration: Risk scores displayed in patient lists, care-team dashboards, discharge planning modules.
Outcomes: Proactive interventions, resource allocation, reduced penalties from readmissions.
2. Clinical decision support enhancement (contextual guidance)
What it does: AI provides treatment suggestions, drug dosing adjustments, and guideline-based reminders contextualized to patient data.
EHR integration: Inline suggestions within order entry and charting; alerts with risk context to reduce alert fatigue.
Consideration: Need for transparent, explainable models and clinician override paths.
3. Natural Language Processing (NLP) for free-text capture
What it does: Converts physician notes, triage text, and discharge summaries into structured problem lists and coded diagnoses.
EHR integration: Auto-populating problem lists, billing codes, and quality measures.
Benefit: Documentation time reduction and improved data quality; supports research and analytics.
4. Automated coding and revenue optimization
What it does: AI recommends accurate billing/coding (ICD/CPT) from encounter data, flagging missed opportunities.
EHR integration: Coding assistant within discharge workflows, audit trails for billing teams.
Outcome: Improved revenue capture and reduced manual coding errors.
5. Intelligent workflow automation (clerical task offload)
What it does: Schedules follow-ups, triages messages, auto-fills forms, routes referrals using rule+ML engines.
EHR integration: Background bots linked to messaging and scheduling modules.
Impact: Frees clinicians for higher value care; reduces administrative backlog.
6. NLP-driven clinical summarization & handoffs
What it does: Produces concise visit summaries and handoff briefs from raw notes and orders.
EHR integration: Handoff screens in inpatient units; discharge summaries for primary care.
Outcome: Safer transitions of care and reduced information omission.
7. Population health analytics & cohort discovery
What it does: AI identifies cohorts (e.g., diabetic patients with poor control) for interventions and clinical trials recruitment.
EHR integration: Population dashboards, automated registry updates.
Impact: Targeted outreach, quality improvement, and public health planning.
8. AI-assisted interoperability mapping
What it does: Maps terminologies and data elements across disparate systems to improve semantic interoperability.
EHR integration: Middleware or EHR modules that normalize incoming data streams (labs, meds).
Benefit: Smoother external data ingestion and less manual reconciliation.
9. Voice recognition and clinical documentation assistants
What it does: Converts clinician dictation into structured notes, highlights action items, auto-suggests orders.
EHR integration: Real-time note creation integrated with problem lists and order sets.
Outcome: Time savings, higher documentation fidelity.
10. Continuous monitoring & early warning from wearables
What it does: Aggregates wearable data (heart rate, glucose) and runs anomaly detection for alerts.
EHR integration: Patient timelines and alert feeds in outpatient monitoring modules.
Utility: Enables remote chronic disease management and reduces acute exacerbations.
Regional Insights in Electronic Health Records (EHR) Market
North America — (USD 10.01 billion in 2024)
Drivers & features
Regulatory incentives: HITECH-era legacy and ongoing HHS actions drive interoperability and patient access.
Provider expectations: Large hospital systems require enterprise EHRs with deep integrations (imaging, lab, revenue cycle).
Vendor maturity: Market consolidation with entrenched vendors handling complex enterprise needs.
Constraints & risks
Complex procurement cycles: Long sales cycles due to capital budgets and committee approvals.
High security/regulatory burden: Strict compliance requirements (privacy, audits) increase operational costs.
Strategic implications
Vendors must deliver enterprise-grade security, deep analytics, and value-based care capabilities to remain competitive.
Europe — (USD 7.15 billion in 2024)
Drivers & features
Policy & interoperability push: EU member states and national strategies emphasize secure data exchange and patient portability.
Diversity: Multiple national health systems create both opportunity (multi-country rollouts) and complexity.
Constraints & risks
Regulatory variance: GDPR and local interpretations create legal complexity for cross-border data flows.
Interoperability challenges: Different coding standards and patient identifiers across countries.
Strategic implications
Vendors who localize regulatory compliance and support multi-language, multi-standard deployments will gain advantage.
Asia-Pacific — (USD 7.15 billion in 2024)
Drivers & features
Heterogeneous maturity: High maturity in countries with strong health budgets; emerging adoption in others.
Telehealth acceleration: COVID-19 catalyzed telemedicine and remote monitoring integration with EHRs.
Constraints & risks
Infrastructure variance: Differences in internet penetration and data centers across countries impede uniform cloud strategy.
Strategic implications
Modular, mobile-first EHR products and partnerships with local integrators are effective go-to-market strategies.
Latin America — (USD 2.288 billion in 2024)
Drivers & features
Modernization trend: Governments and private systems upgrading core infrastructure.
Clinical need: Large urban hospitals drive initial demand.
Constraints & risks
Funding limitations: Capital constraints slow large-scale EHR rollouts; preference for lower-cost or open-source systems.
Strategic implications
Affordable SaaS models and phased implementation approaches are pragmatic market entry routes.
Middle East & Africa — (USD 2.002 billion in 2024)
Drivers & features
Strategic investments: Wealthier ME countries (UAE, Saudi) invest in advanced health IT and EHR modernization.
Leapfrogging potential: Some systems can adopt cloud and mobile solutions rapidly.
Constraints & risks
Low-income country barriers: Poor connectivity, workforce shortages, and distrust of digital systems impede adoption.
Strategic implications
Mixed approach: premium enterprise solutions for wealthy GCC markets; lightweight, offline-capable systems for lower-income regions.
Market Dynamics in Electronic Health Records (EHR) Market
A. Primary market drivers (detailed)
Clinical workload & acuity: Rising chronic disease prevalence and hospitalizations push demand for robust EHRs to manage complex care.
Regulatory requirements & incentives: Laws mandating electronic records and promoting interoperability increase adoption.
Cloud economics: Lower total cost of ownership and faster deployment catalyze cloud migration.
Quality & safety focus: Need for clinical decision tools, documentation, and auditability drives purchases.
B. Key restraints (detailed)
Cost & implementation complexity: Upfront costs, long implementation times, and the need for clinician training inhibit some buyers.
Change management: Clinician resistance due to perceived usability issues or increased documentation time.
Infrastructure gaps: Poor connectivity in low-income regions reduces feasibility of cloud-first strategies.
Data security concerns: Fear of breaches delays adoption or forces expensive security investments.
C. Opportunities (detailed)
AI-enabled modules: Upsell and product differentiation via AI/analytics modules (predictive care, population health).
Post-acute & home care expansion: Growth in home health creates demand for interoperable EHRs that follow the patient.
Interoperability services & HIEs: Vendors can monetize data exchange, normalization, and HIE participation.
Patient engagement products: Portals, remote monitoring, and telehealth integrations open new product lines.
D. Challenges (detailed)
Vendor lock-in & migration costs: Legacy systems create frictions for switching vendors; migration complexity is a barrier.
Standards fragmentation: Multiple clinical terminologies and message formats complicate true interoperability.
Talent shortage: Skilled IT and informatics personnel are scarce in many regions, slowing deployments.
Top 10 Companies in Electronic Health Records (EHR) Market
1. Epic Systems Corporation
Products: EpicCare, MyChart, modules for inpatient/outpatient workflows.
Overview: Market leader in large U.S. health systems and academic medical centers.
Strengths: Deep enterprise deployments, strong interoperability within Epic community, patient portal (MyChart) adoption, extensive implementation and support capabilities.
Strategic notes: High switching costs for customers; strong role in value-based care analytics.
2. Allscripts Healthcare Solutions, Inc.
Products: Sunrise (hospital), TouchWorks (ambulatory), CareInMotion (population health).
Overview: Offers both acute and ambulatory solutions with emphasis on open architectures.
Strengths: Flexibility & customizability; integration-friendly; appeals to health systems seeking tailored solutions.
3. McKesson Corporation
Products: Horizon Clinicals (legacy), integrated supply chain & EHR solutions.
Overview: Large health IT and supply chain player with EHR offerings tied to broader operations.
Strengths: Strong integration between pharmacy/supply chain and clinical systems; enterprise scale.
4. GE Healthcare
Products: Centricity family (EHR + imaging integrations).
Overview: Combines diagnostic imaging and EHR capabilities—valuable for hospitals where imaging is central to care.
Strengths: Deep integration of PACS and clinical systems; strength in diagnostic workflows.
5. NextGen Healthcare, Inc.
Products: NextGen Office, NextGen Enterprise.
Overview: Strong focus on ambulatory and specialty practices (including recent podiatry blueprints).
Strengths: Tailored ambulatory workflows, configurable templates, and SMB clinic fit.
6. Athenahealth
Products: athenaClinicals, athenaOne suite (cloud-based).
Overview: SaaS-first model aimed at efficiency and payer-provider integration.
Strengths: Cloud-native, subscription economics, strong revenue cycle and interoperability features.
7. OpenEMR
Products: Open-source EHR platform (community-driven).
Overview: Widely used as low-cost customizable platform in resource-limited settings.
Strengths: Cost-effectiveness, high customizability, community support; attractive for smaller clinics and NGOs.
8. Siemens Healthineers AG
Products: Integrated EHR solutions + imaging/diagnostics.
Overview: European-rooted vendor integrating clinical workflows with diagnostics.
Strengths: Strong presence in EU hospitals, deep technical integration with imaging and diagnostics.
9. eClinicalWorks
Products: Ambulatory EHR + telehealth modules.
Overview: Large ambulatory vendor with patient engagement tools.
Strengths: Telehealth integration, patient portal, strong midsize clinic footprint.
10. Medical Information Technology, Inc. (MEDITECH)
Products: Expanse EHR platform, cloud and on-prem solutions.
Overview: Long-time EHR vendor serving hospitals and health systems; partnership with Canada Health Infoway for e-prescribing.
Strengths: Proven hospital workflows, e-prescribing integrations, strong regional partnerships (e.g., Canada).
Latest Announcements
1. U.S. HHS — Patient access & policy refinement (Jan 23, 2024)
What: HHS working to improve policies to enhance patient access to health information.
Why it matters: Policy changes push vendors to ensure APIs/portals expose patient data securely; affects product roadmaps toward better patient-facing features and consent handling.
2. ONC & HITAC involvement (Oct 2023 / Jan 18, 2024)
What: Health Information Technology Advisory Committee (HITAC) advising ONC on interoperability and privacy standards.
Why it matters: Vendors must align roadmaps to recommended standards; hospital procurement will favor vendors that support ONC-recommended APIs and privacy controls.
3. Prime Healthcare — athenahealth implementation (CEO Susan Albano)
What: Prime Healthcare reports benefits from athenahealth adoption—improved provider engagement and payer data access.
Why it matters: Case example for successful cloud EHR implementation improving value-based care operations and analytics.
Recent Developments
July 2023 — NextGen & American Podiatric Medical Association (APMA)
What: NextGen enhanced “NextGen Office” with podiatry blueprints addressing diabetes and injuries.
Why it matters: Specialty-specific content increases adoption in niche practices — demonstrates vendors’ strategies to capture specialty markets with tailored templates & workflows.
June 2023 — CPSI & Mid Coast Health System expansion
What: CPSI expanded EHR, AR, and IT-managed services across multiple Mid Coast hospitals in Texas.
Why it matters: Example of regional rollouts where community hospitals standardize on one vendor for EHR + revenue cycle + managed services, capturing recurring services revenue.
May 2023 — MEDITECH & Canada Health Infoway (Prescribell)
What: MEDITECH linked Expanse EHR with national e-prescribing service.
Why it matters: National e-prescribing ties improve medication safety, streamline renewals and cancellations, and show the benefit of national-level integrations for EHR vendors.
Feb 2023 — King’s College Hospital Dubai & Oracle Cerner cloud migration
What: Migration to Oracle Cerner using Oracle Cloud for EMR modernization.
Why it matters: Major hospital cloud migrations are proof cases for enterprise cloud EHR viability in large, complex settings and for cross-border deployments.
Segments Covered
By Type (Acute, Post-Acute, Ambulatory)
Acute (Largest):
Use-cases: ED, ICU, surgical, inpatient charting.
Requirements: High availability, integration with critical care devices, medication administration records (MAR).
Vendor focus: Enterprise modules, ICU workflows, imaging and lab tight integrations.
Post-Acute (Fastest-growing):
Use-cases: Home health, skilled nursing, rehabilitation, long-term care.
Requirements: Mobility, offline capability, care-coordination features, simplified documentation for non-specialist staff.
Market trend: Aging populations and discharge-to-home strategies increase demand.
Ambulatory:
Use-cases: Primary care, specialty clinics, outpatient surgery centers.
Requirements: Scheduling, quick documentation, telemedicine, and integrated revenue cycle for smaller practices.
Vendor focus: Lightweight, fast-to-implement modules; specialty templates.
By Product (Web server-based, Client-server-based)
Web Server-Based (Leader):
Characteristics: Cloud-hosted, multi-tenant models, frequent updates.
Advantages: Scalability, remote access, reduced internal IT.
Customer fit: Health systems preferring OpEx and fast innovation cycles.
Client-Server-Based (Fastest growth):
Characteristics: Locally hosted, more customization, perceived as more secure by some institutions.
Advantages: Greater control, customization for specialized workflows, potential cost benefits in certain contexts.
Customer fit: Organizations with strict localization or regulatory constraints or wanting local control.
By End-User (Hospitals, Clinics, Ambulatory services, Specialty centers)
Hospitals:
Requirements: Unified inpatient + outpatient record, enterprise governance, high-resilience infrastructure.
Value drivers: Efficiency gains, compliance, analytics for outcomes & reimbursement.
Clinics & Ambulatory Services:
Requirements: Low overhead, speed of patient throughput, simplified documentation.
Value drivers: Productivity improvements and patient portal access.
Specialty Centers:
Requirements: Specialty-specific templates, procedure documentation, integration with specialty devices.
Value drivers: Improved specialty workflows and better patient outcomes in niche areas.
Top 5 FAQs
1. What is the market size of the EHR industry in 2024?
Answer: The market is USD 28.60 billion in 2024. This includes software licenses/subscriptions, services (implementation and managed services), and infrastructure support. The value is driven by hospital enterprise deployments, ambulatory adoption, and growing cloud migration.
2. What is the expected CAGR of the EHR market from 2024–2034?
Answer: 4.32% CAGR. This signals steady, sustainable growth as mature markets continue upgrades and developing markets scale adoption; growth is supported by cloud adoption, policy incentives, and post-acute care expansion.
3. Which EHR product type leads the market and why?
Answer: Web server-based (cloud) EHRs led the market in 2023 (48% share) because cloud models provide scalability, remote access, and lower internal IT burden. They enable quicker product updates and easier integrations with analytics and AI modules.
4. Which segment is the fastest-growing and why?
Answer: Post-acute segment — driven by aging populations, home health services expansion, and the need for seamless transitions from inpatient to outpatient/home care which require interoperable, mobile-capable EHR solutions.
5. Who are the major EHR vendors and what differentiates them?
Answer: Major vendors include Epic, Allscripts, McKesson, GE Healthcare, NextGen, Athenahealth, OpenEMR, Siemens Healthineers, eClinicalWorks, and MEDITECH. Differentiators: scale and enterprise focus (Epic), cloud-first economics (Athenahealth), vertical integrations with imaging/diagnostics (GE/Siemens), cost-effectiveness and flexibility (OpenEMR), and ambulatory specialty focus (NextGen, eClinicalWorks).
Check out the details below
Access our exclusive, data-rich dashboard dedicated to the healthcare market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Immediate Delivery Available | Buy This Premium Research @ https://www.towardshealthcare.com/price/5103
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest